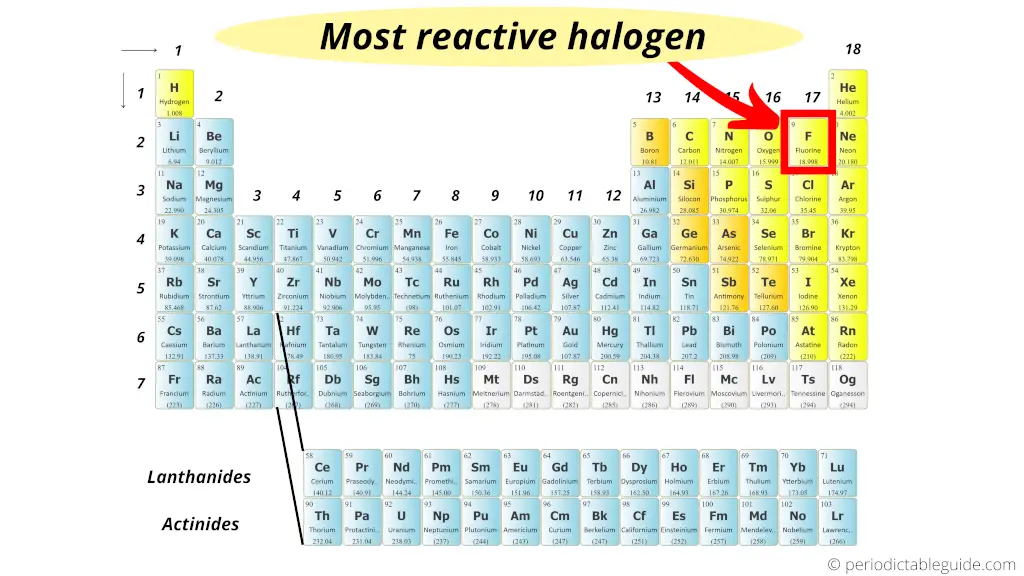  
They have one electron found in their outermost shell. They are known as group $1$ elements and are found in the s-block of the periodic table. We have to know that members present in alkali metal family are potassium $\left( K \right)$, sodium $\left( \right)$. The reason why they are highly reactive is because of the electronic configuration. We can say that the family which has the most reactive metals in the periodic table is alkali metals. As such, as you increment the size of atoms of metals in a group, their reactivity likewise rises. The reactivity series of metals is a chart listing metals in order of decreasing reactivity. These electrons are simpler to eliminate, so the atoms promptly form chemical bonds. As you drop down a column or group of the periodic table, there would be an increase in size of the atomic radius.įor the metals, this implies the valence electrons turn out to be farther away from the nucleus which is positively-charged. A component that is exceptionally electronegative, like fluorine, has an incredibly high attraction for holding electrons.Ĭomponents at the far edge of the range, like exceptionally reaction metals cesium and francium, promptly form chemical bonds with electronegative particles. The alkali metals are so called because reaction with water forms alkalies (i.e., strong bases capable of neutralizing acids). We have to know that Reactivity is a proportion of how possible a chemical species is to partake in chemical reactions to produce chemical bonds. alkali metal, any of the six chemical elements that make up Group 1 (Ia) of the periodic tablenamely, lithium (Li), sodium (Na), potassium (K), rubidium (Rb), cesium (Cs), and francium (Fr). These are, by period on the periodic table and. The arrangement is a chart that rundowns components as per how promptly the metals displaces hydrogen in chemical reaction. There are thirty six that have no radioactive isotopes found in nature except as a result of human activity or, very rarely, as decay products. Furthermore, sodium will readily bond with any. This element is in the alkali metals family, and it has only one valence electron. The general reaction for alkali metal hydrides and water is given below: (4) M H ( s) + H 2 O ( l) M ( a q) + + O H ( a q) + H 2 ( g) This reaction can be. Sodium, atomic symbol Na, has an atomic number of eleven, and lies in group 1 on the periodic table, below lithium and to the left of magnesium. In this case, however, hydrogen gas is produced with the metal hydroxide. We can therefore confirm their order of reactivity (from most reactive to least) as: potassium, magnesium then zinc.Hint: We have to know that one can utilize the reactivity of metals series to foresee which metal will be the most reactive and to look at the reactivity of various metals. Similarly to the Group 1 oxides, the hydrides of the Group 1 elements react with water to form a basic solution. We’d expect the potassium to react explosively (in reality you would never do this in a school laboratory as it is too dangerous), while the magnesium would bubble vigorously and the zinc would form bubbles very slowly. Let’s say we carry out the experiment described above by adding potassium, magnesium and zinc into test tubes containing dilute sulfuric acid. The faster the bubbles are given off, the faster the rate of reaction and the more reactive the metal. Reactive nonmetals are a group of chemical elements that are located in the upper right-hand corner of the periodic table.This group includes 11 elements, namely hydrogen (H), carbon (C), nitrogen (N), oxygen (O), fluorine (F), phosphorus (P), sulfur (S), chlorine (Cl), selenium (Se), bromine (Br), and iodine (I). The alkali metals lithium, sodium, potassium, rubidium, cesium, and francium constitute group 1 of the periodic. Review the Periodic Table of the Elements in other formats in Appendix A (credit: Chemistry (OpenStax), CC BY 4.0). The bubbles are hydrogen gas and can be confirmed using a lit splint, which will produce a ‘squeaky pop’ when the hydrogen burns. From the table, note that the most reactive metal on the periodic table is cesium. Figure 3.2g: The periodic table organizes elements with similar properties into groups. 
It is important that each metal has the same surface area because this will affect the rate of reaction.Ĭount the number of bubbles produced in a given time. Add equal volumes of dilute hydrochloric acid or dilute sulfuric acid into a series of test tubes then add a equal mass of metal to each test tube.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
